PRODUCED
WATER REPORT
Understanding naphthenate salt issues in oil
production
These
troublesome salts, formed when oil-soluble naphthenic acids come
in contact with high-pH produced water, cause tight emulsions,
ragged interfaces, organic deposition and, ultimately, separation
difficulties.
R. Anthony Rodriguez and Sen
J. Ubbels, Champion Technologies
Various naphthenic acids are present in nearly all crude oils in different
forms. Problems occur when the naphthenic acids come in contact with
high-pH connate waters. The problems start at the oil-water interface
and can cause organic deposition, impede oil-water separation and ultimately
lead to poor water quality.
This article reviews the characterization of, problems caused by and
remediation of naphthenate salt issues in the upstream environment,
drawing information from direct experience and literature.
INTRODUCTION
Crude oil is a complex mixture of 10,000-100,000
distinct compounds.1 Typically, the components are organized into
the well-known fractions of Saturates, Aromatics, Resins and Asphaltenes
(SARA). Included in these fractions are oil-soluble naphthenic acids.
At reservoir conditions, the acids are contained in the oil phase
and demonstrate low interfacial activity.2 As the pressure cascades
up through the flowline and ultimately into the topside separation
equipment, depressurization occurs.
Each fraction is soluble in its respective
media, and all are held together in the complex system that is crude
oil. The mixture is stable at reservoir conditions, but perturbation
can change the fluid’s
dynamics enough to cause complex operational issues. For example, asphaltenes
are stable at reservoir conditions as a colloidal suspension dispersed
with resins and solubilized with aromatics.3 As fluid moves out of
the reservoir and into the tubing, its pressure drops, and the lighter
components’ relative volume increases, decreasing the colloid
solubility. This causes the asphaltenes to become unstable and sometimes
sticky, resulting in deposition.
Similarly, the naphthenic acids in crude oil, regardless of their
structure, reside harmlessly in the oil phase under reservoir conditions.
The depressurization that occurs as oil moves out of the reservoir
and up the production tubing can cause the pH of system water to increase,
resulting in a salt with ions from the water and naphthenic acids from
the oil. An anionic hydroxyl is necessary to create the salt, as is
illustrated in the equation:4
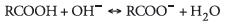
The migration of the anionic naphthenic acid to the interface allows
the reaction with metal ions, yielding the troublesome naphthenate
salts.
Specific issues that directly result from naphthenate salts include
very tight emulsions, ragged interfaces, high calcium levels in export
crude oil and organic deposition. Difficulties in treating produced
water and other indirect problems may also result.
ORGANIC DEPOSITION
An offshore facility at field A produced a crude oil-water combination
that, when depressurized in the first stage separator, created organic
deposits at the oil-water interface.5 The deposits plugged the baffles
in the separator, as well as in water treatment equipment. The plugging
of the separator caused level control problems, which resulted in water
being carried over into dehydration stages. Oil was also carried over
into the water and overwhelmed the water treatment equipment, resulting
in poor water quality.
To control the deposits, the pH was adjusted down to the acid range
using acetic acid upstream of the first stage separator. The acetic
acid was not completely successful in achieving remediation of the
deposit, because strong corrosion and logistics prevented injection
of sufficient acid.
Specifically, the use of acids for naphthenate control presents logistical
issues with regard to safety and storage. Handling corrosive materials
in large quantities presents safety issues for the operations personnel.
Chemical storage offshore is always at a premium, as the space available
is not flexible. The use of naphthenate inhibitors in this case reduced
the amount of acid used, thus simplifying logistical issues. Not until
a naphthenate inhibitor was included in the remediation effort was
the organic deposition completely controlled.
CHARACTERIZATION
A great deal of research has been pursued
to characterize the naphthenic acids responsible for organic deposition,
Fig. 1.6 Thousands of naphthenic acids have been identified, and
with each change in structure comes a change in the acid’s
interfacial activity. The structure may vary by the number of rings,
saturation or, in some cases, the number of carboxylic groups.
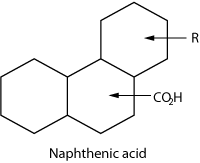 |
Fig.
1. The characterized standard naphthenic acid
shown here is of a polycyclic species, which is not
considered likely to be responsible for organic depositions. |
|
Naphthenic acid species have been characterized using Gas Chromatography
and Mass Spectrometry (GC-MS), two-dimensional GC-MS, Fourier-Transform
Ion Cyclotron Resonance (FT-ICR) mass spectrometry and Nuclear Magnetic
Resonance (NMR) spectroscopy. Recently, the naphthenic acid responsible
for solids deposition was identified as an archaeal C80 isoprenoid,
also known as ARN.7 This acid was identified as a tetra-acid, with
four carboxylic groups, Fig. 2.
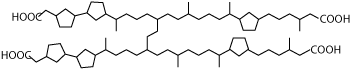 |
Fig.
2. The naphthenic acid responsible for solids
deposition was recently identified as an archaeal C80
isoprenoid, also known as ARN.8 This acid was identified
as a tetra-acid, with four carboxylic groups. |
|
These groups afford the molecule unusually high reactivity. The four
carboxylic groups tend to create polymeric salt when they are coordinated
with divalent metal ions. This weaved polymeric-like structure yields
a very sticky deposit that hardens upon contact with air. Once a deposit
sets, it can only be dissociated by using a mixture of hot acid and
aromatic solvent in laboratory conditions. A remediation using mineral
acids in the field would typically be too severe from a safety and
corrosion perspective.
INTERFACIAL RAG LAYER
Field B is another offshore facility that did not experience organic
deposition, but experienced an unresolved webby interface that impeded
water treatment. The overboard water produced had very high oil and
grease counts, although the sales oil tank had very dry, clean oil.
A total system survey found the interface at four sample-point levels
in the Free-Water KnockOut (FWKO). The dirty interface carried over
to the water treatment hydrocyclone and float cell. The water treatment
equipment was overwhelmed, resulting in dirty overboard water.
Grind-out tests of the individual wells showed that a few wells produced
a pad layer, which the operator was unable to break using a standard
slugging compound. The addition of a naphthenate inhibitor into the
subsequent grind-outs cleared up the interface. Ultimately, a demulsifier
not containing a naphthenate inhibitor was applied at the FWKO. It
was able to remove the dirty interface from the produced water. After
the new demulsifier was plant tested, the FWKO was sampled, and unresolved
interface was only found at one level on the FWKO. The carryover to
the water treatment equipment had been reduced, and the overboard oil
and grease counts met or exceeded specifications. In this case, the
use of the demulsifier package was enough to overcome the emulsifying
characteristics of the naphthenate salts.
TIGHT EMULSIONS, REMEDIATION AND PREDICTION
Naphthenic acids are interfacially active, which affords them unique
stabilization properties, even in a slightly acidic environment of
pH ~ 6.5. As the pH goes up, though, the interfacial activity goes
up, and salt formation becomes more likely, Fig. 3.9 When an added
acid brings the pH down, the emulsion problems may decrease in severity.
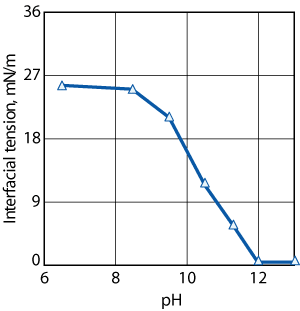 |
Fig.
3. In this model system of water and acidic
oil, the interfacial tension changed as the pH increased.
The change in interfacial tension was correlated to
the formation of RCOO- ions. As the pH increased,
more RCOO- ions were formed, and the interfacial
tension decreased until the interface was saturated.
This data follows field observations about the resolution
of the emulsion, and the pH in the system. |
|
The structure of the acid strongly affects the interfacial tension
and the rate at which the interfacial tension changes with a change
in pH. In the model system shown in Fig. 3, it is not clear how even
in regions of moderately low pH, the naphthenic acids are still active.
In the field, even at pH = 6, problems at the interface still occur.
Field C produced a very stable emulsion that had the consistency of
muddy water from some locations, and viscous, stable emulsions from
other locations. The operator was unable to break the stable emulsion
with a typical non-ionic demulsifier; only a highly acidic demulsifier
or acid-demulsifier combination products were effective in treating
it. The acid portions included the use of DoDecylBenzene Sulfonic Acid
(DDBSA), acetic acid and mineral acids. The addition of a naphthenate
inhibitor to a demulsifier allowed the operator to minimize the use
of acids and increased the effectiveness of the oil-water separation.
The naphthenate salts in field C, and in most systems during the past
20 yr, have been treated using acids of some kind. The inhibiting mechanism
of a pure acid is likely to increase the interfacial tension, thus
better defining the interface. As pH decreases with the addition of
acid, the dissociation of the naphthenic acid to the anionic functional
group is decreased. As the occurrence of the anionic species decreases,
the surface tension goes up and the surfactancy of the naphthenic acid
goes down. This will limit the tightness of the emulsion or the interface,
but these issues can continue to exist into the acidic range.
Another method of controlling naphthenate salts is the use of specialty
surfactants known as naphthenate inhibitors. These surfactants use
the mechanism of interfacial crowding to inhibit salt formation, Fig.
4. The surfactant is more interfacially active than naphthenic acid,
which reduces the probability of a reaction between the salt and the
acid.10 If the naphthenic acid is not allowed to reach the interface,
it cannot cause solid deposition or emulsion. In our experience, surfactant
treatments, in the form of demulsifiers, naphthenate inhibitors, or
acid combinations, are much more effective at much lower dosages than
acid alone.
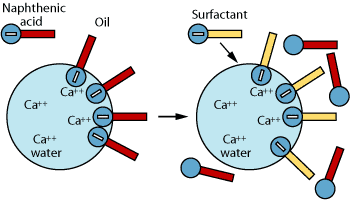 |
Fig.
4. Specialty surfactants known as naphthenate
inhibitors use the mechanism of interfacial crowding
to inhibit salt formation. |
|
Due to the varying structures of naphthenic acids, the treatments
and injection points may vary greatly. For example, the injection point
in a fluid with tetrameric naphthenic, or ARN, acid is much more critical
than in a fluid with monoacid-type naphthenic acid. In the case of
the ARN acid, the treatment must be upstream of the point where polymeric
salt is formed because dissociation after the formation is very difficult.
The converse is true with a monoacid: After salt formation, dissociation
may still be simple, providing some flexibility to the treatment plan.
There are case histories that require downhole treatments because of
the difficulty of dissociation.
Until recently, naphthenate salt problems have been dealt with reactively,
and not planned for in the design phase. Although it is possible to
predict high pH, there are no time-tested early indicators of the presence
of the naphthenic acids that would cause a problem. The problem can
be identified in field as the presence of a strong rag layer or by
the solubility of an organic deposit. Analytical techniques have evolved
to a point where certain naphthenic acids can be identified in problem
fields. For example, the ARN acid has been identified at 1,200 amu
via mass spectroscopy. The structure has been plotted with the aid
of NMR. As more is learned about naphthenate salt problems, it will
become possible to better predict and thus plan for such issues.
Champion Technologies has initiated research
to understand emulsion tendency trends associated with troublesome
oils. Using the pendant-drop “tracker” technique,
one can plot interfacial tension vs. pH, Fig. 5. The technique uses
the Laplace equation for bubble geometry change. The collection of
this data is empirical at present, but it may shed light on trends
for surfactant use. These trends may also predict ragged interfaces
or even deposits.
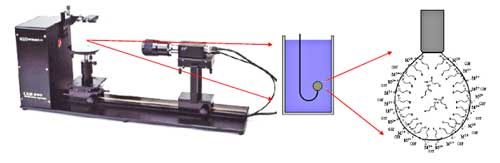 |
Fig.
5. The pendant-drop “tracker” technique
is used to plot interfacial tension vs. pH, Courtesy
of the Norwegian University of Science and Technology. |
|
CONCLUSION
Naphthenic acids of varying structures exist in nearly all crude oils,
but remain inert in upstream conditions without a perturbation in the
system. Certain perturbations in the system may lead to interfacially
active naphthenate salts.
The structure of a given naphthenic acid dictates its interfacial
activity. More interfacially active naphthenic acids in slightly acidic
to basic pH waters may form salts that, in turn, form tight emulsions
or rag layers. Some naphthenic acids may coordinate with divalent metal
ions to form polymeric salts, which lead to organic deposition. The
naphthenate salts may cause direct problems such as emulsions. Indirect
problems resulting from the formation of emulsions or organic deposition
include oil carryover into water treatment systems and water carryover
into oil systems.
Methods for remediation of naphthenate
salt issues include the use of acids and/or specialty surfactants
(i.e., naphthenate inhibitors). The best treatment at present is
only reactive, as solid modeling methods in the early planning stages
do not exist. Developing solutions on site with fresh fluids is critical
to the success of a treatment program. The use of a standardized
naphthenate inhibitor and demulsifier kit simplifies the process.
Predictive tools will likely progress as the physiochemical properties
of different naphthenate species become better understood. 
LITERATURE
CITED
1 Klein, G. C. et al., “Use of saturates/aromatics/resins/asphaltenes
(SARA) fractionation to determine matrix effects in crude oil analysis
by electrospray ionization fourier transform ion cyclotron resonance
mass spectrometry,” Energy & Fuels, 20, March 2006, pp.
668-672.
2 Ubbels,
S. J. and M. Turner, “Diagnosing and preventing
naphthenate stabilized emulsions during crude oil processing,” presented
at the 6th Petroleum Phase Behaviour and Fouling Conference, Amsterdam,
June 19-23, 2005.
3 Klein et al.
4 Arla,
D. et al., “Influence of pH and water content on
the type and stability of acidic crude oil emulsions,” Energy & Fuels,
21, May 2007, pp. 1337-1342.
5 Hurtevent,
C. and S. Ubbels, “Preventing naphthenate stabilised
emulsions and naphthenate deposits on field producing acidic crude
oils,” presented at the 2006 SPE International Oilfield Scale
Symposium, Aberdeen, Scotland, May 30-June 1, 2006.
6 Qian,
K. et al., “Fundamentals and applications of electrospray
ionization mass spectrometry for petroleum characterization,” Energy & Fuels,
18, November 2004, pp. 1784-1791.
7 Lutnaes,
B. F. et al., “Archaeal C80 isprenoid tetraacids
responsible for naphthenate deposition in crude oil processing,” Organic & Biomolecular
Chemistry, 4, Feb. 21, 2006, pp. 616-620.
8 Brandel,
O. et al., “Interfacial behavior of C80 tetrameric
naphthenic acids responsible for naphthenate deposition in crude
oil processing,” presented at the 7th Petroleum Phase Behaviour
and Fouling Conference, Ashville, North Carolina, USA, June 2006.
9 Arla et al.
10 Hurtevent and Ubbels.
|
THE AUTHORS |
|
Anthony
Rodriguez is a flow assurance/flow management specialist working
out of Champion Technologies’ main Tech Center in Fresno,
Texas. His primary functions are project and customer support
of the wax, asphaltene, defoamer, drag reducer, and calcium
naphthenate product lines for the Western Hemisphere. |
|
 |
Sen
Ubbels is the Phase Separation section manager for
Champion Technologies in the Eastern Hemisphere, working out
of the Tech Center in Delden, the Netherlands. His primary
functions are as manager of the demulsifier, water clarifier,
defoamer and calcium naphthenate product lines and personnel.
Mr. Ubbels pioneered the calcium napthenate inhibitor product
line for Champion. |
|